
Institutional Healthcare Risk Architecture and Federal Safety Oversight .
- Our work is informed by over 20 years of experience across provider, payer, regulatory, and industry settings. We understand how safety documentation, risk analyses, and corrective actions are evaluated by auditors and regulators, and structure findings accordingly.
- This experience includes work within academic medical centres, federal healthcare oversight, multi-specialty medical practices, trauma hospitals, and medical device environments, followed by advanced academic training and published research in patient safety and systems design
- Enginova Health Systems works within existing healthcare technology environments to support evidence-based analysis, documentation review, and systemic risk assessment.
What we do —
- Pre-Audit Risk Identification: risk domain mapping, evidence readiness, audit response preparation
- Control Effectiveness & Evidence Mapping: traceable documentation aligned with regulator and payer review
- Human Factors & Systems Analysis: workflow, handoffs, usability, cognitive load, and oversight mechanism review
- CAPA & Governance Reinforcement: corrective actions with owners, timelines, and monitoring indicators
Deliverables
- Risk register and prioritized risk pathways
- Control effectiveness review
- Audit-ready evidence mapping (traceability)
- CAPA plan with owners, timelines, and monitoring indicators
- Executive summary suitable for governance review
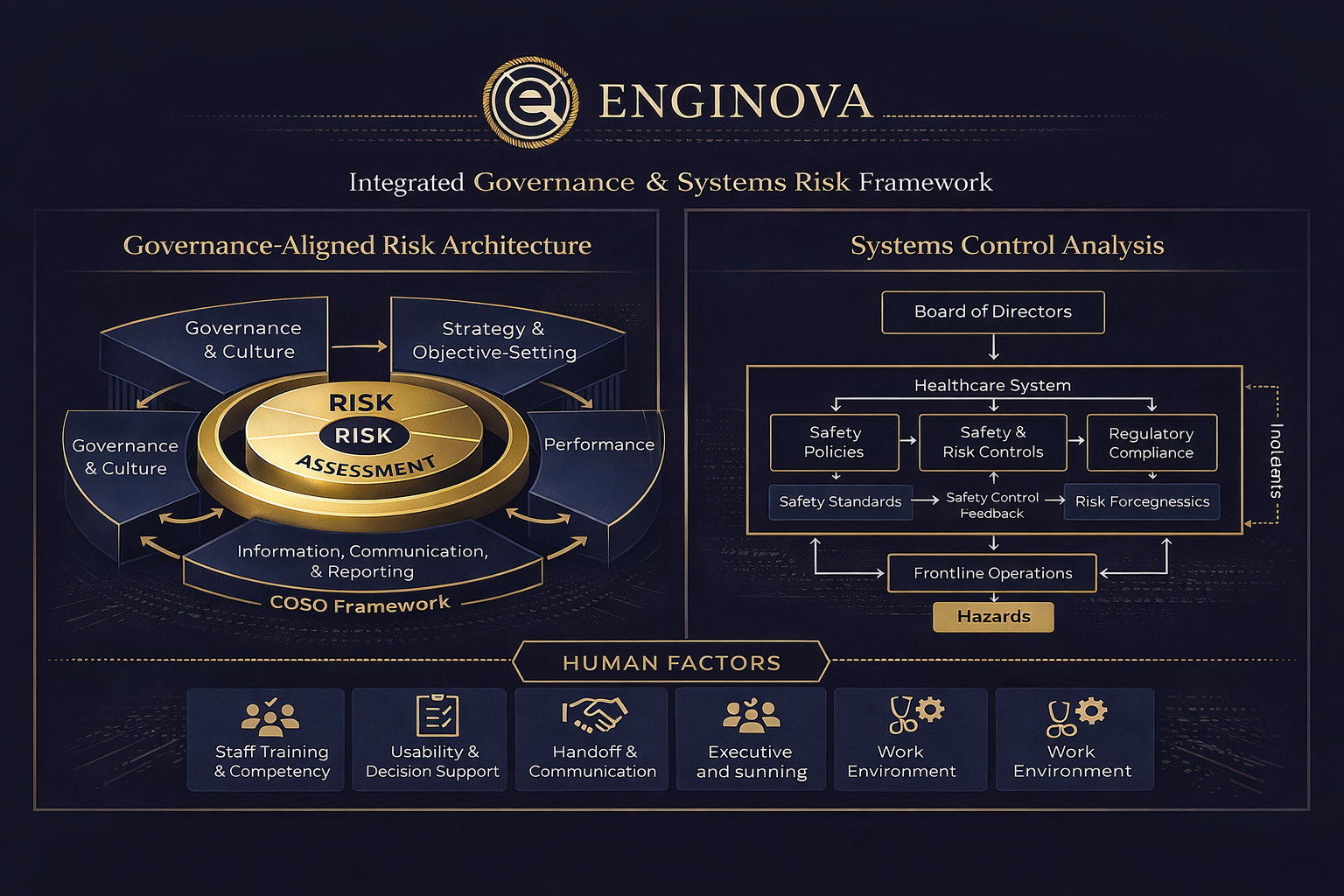
Human Factors & Systems Analysis
- We evaluate how risk emerges from interactions between people, workflows, technologies, and oversight controls.
- Using systems analysis and human factors engineering, we examine handoffs, decision pathways, usability constraints, and governance mechanisms to identify vulnerabilities not visible through incident review alone.
- Findings are documented as traceable risk pathways linked to control weaknesses and practical corrective actions.
- Workflow and handoff risk mapping
- Human–system interaction and usability constraints
- Oversight and control effectiveness gaps
- Risk pathway prioritisation (severity × likelihood)
PRE-AUDIT & RISK IDENTIFICATION PHASE
- Aligns risk documentation with CMS, payer, and regulatory review expectations.
- Produces traceable evidence mapping showing how risks are identified, controlled, monitored, and sustained over time.
Sustainable CAPA & Governance Reinforcement
- Risk reduction requires corrective action that is owned, monitored, and sustained. We translate findings into proportionate CAPA with clear owners, timelines, and effectiveness indicators suitable for governance review.
- Our approach focuses on control durability—preventing safeguards from eroding over time.
- CAPA actions with owners and deadlines
- Monitoring indicators and effectiveness checks
- Governance reporting structure (what gets reviewed, when, by whom)
- Sustainability review to prevent control drift
Audit-Focused Safety & Compliance Advisory
- Our work is informed by over 20 years of experience across provider, payer, regulatory, and industry settings. We understand how safety documentation, risk analyses, and corrective actions are evaluated by auditors and external reviewers.
- We assess not only compliance, but system resilience — how organizations anticipate, monitor, respond to, and adapt to emerging risk conditions.
Confidential Advisory Engagement
Organizations requiring independent, audit-aligned safety risk review may submit a confidential inquiry for consideration.
Independent Safety Risk Review & Audit Support
- We provide independent, third-party safety risk reviews designed to support internal governance, regulatory inquiry, and external audit preparation.
- Our assessments focus on identifying systemic vulnerabilities, evaluating control effectiveness, and prioritising high-severity risk pathways requiring immediate oversight.
- Engagements are structured to produce objective findings, clearly documented assumptions, and proportionate recommendations suitable for board-level and compliance review.
- Typical engagement outputs include:
- Structured risk findings report
- Prioritised high-severity risk register
- Executive summary for governance review
- Recommendations aligned with existing control frameworks
Control Effectiveness & Evidence Mapping
- Risk register and control inventory
- Evidence mapping (policy → control → monitoring → owner)
- Findings written for audit review and governance action
- Executive-ready summaries and CAPA traceability
